AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Meddra coding guidelines pdf4/7/2024 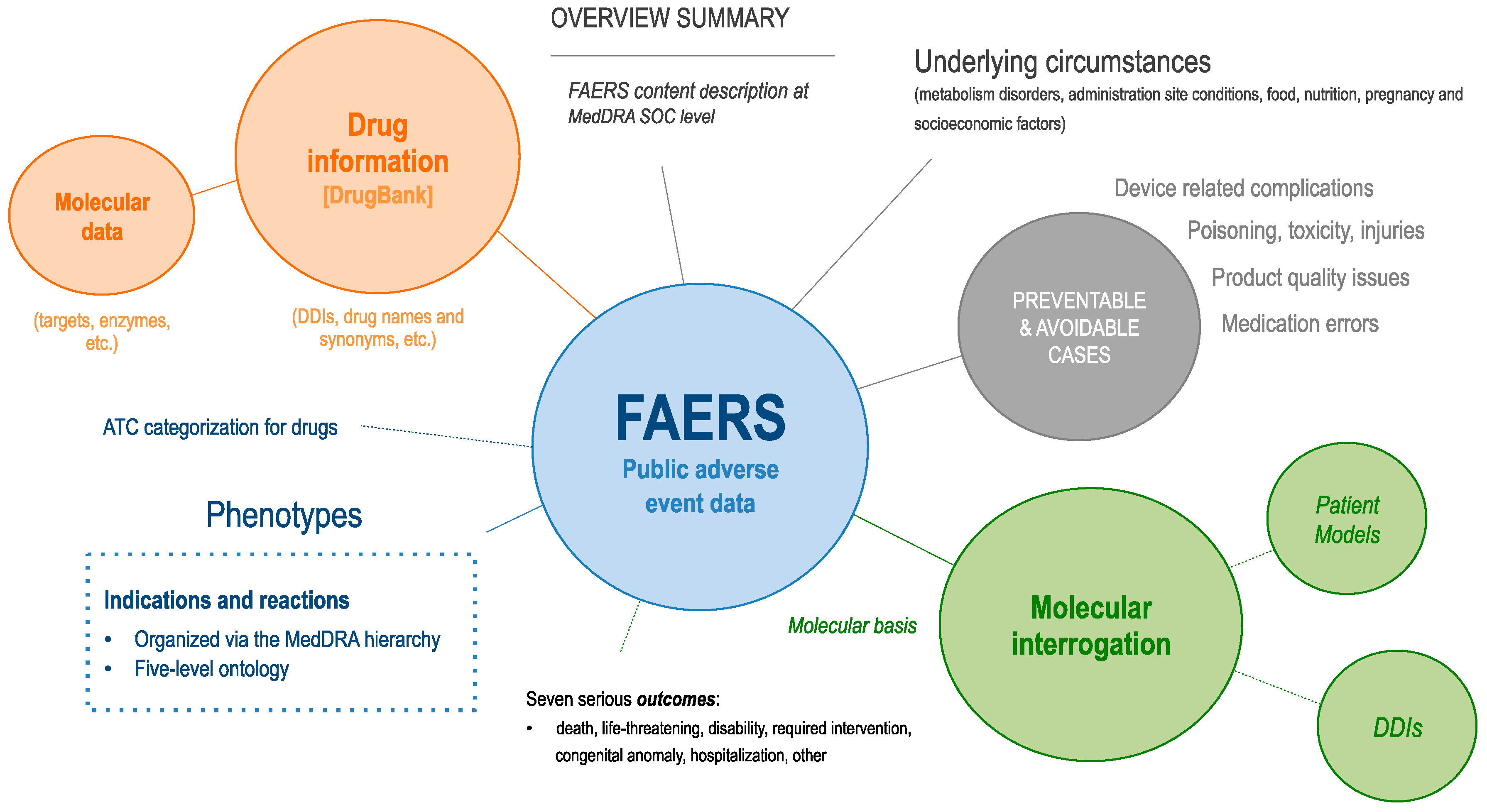
In some studies, patients can contact investigators by phone in other studies, the symptoms may not be recorded before the next visit (which might be weeks ahead). A patient in a trial may experience ‘something’. Readers of medical journals need to be aware of these issues in order to appraise published study reports critically.īefore harms are reported (or not reported) in a published paper, many decisions have been made. The process of condensing thousands of pages of data on adverse events from clinical trials to tables in regulatory submissions and summaries in papers and product labeling is complex and involves many assumptions and choices. Throughout a clinical trial, adverse events, including harms of the drug, are monitored and recorded for the purposes of patient safety, regulatory requirements, and developing a safety profile of the drug. All drugs have unwanted effects and reliable information about these effects is important. 
The decision to prescribe a drug is based on the balance between the drug's benefits and harms.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
